Studies have shown that multi-stage cell lineages, which include stem cell and multiple progenitor cell stages, are under feedback regulation from different growth factors. The existence of regulation is critical for hemostasis of tissue growth during embryonic development and generation of a robust spatial stratification. Regulation is particularly important for rapid regeneration when death of a large number of cells is induced.
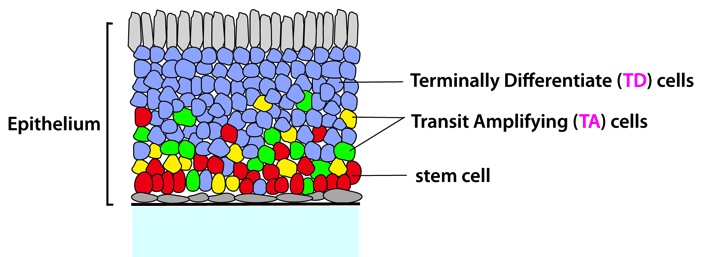
Figure: A cartoon of general epithelia and relative locations of cells at different lineage stages (red, stem cells; yellowand green, TA cells, blue, TD cells). The connective tissue underlying the basal lamina is stroma. (from ref.1)
In our studies, ODE models have been presented for the cell lineages and we have systematically studied the conditions of existence, uniqueness and stability of steady state of the system with an application to the olfactory epithelium. Our analysis shows how negative feedbacks enhance the stability and robustness of the system. To investigate the spatial dynamics of different types of cells in lineages, we extended the non-spatial ODE model by including growth of tissue using a PDE model with moving domain. We found that inter-regulation among different growth factors are responsible for developing layers of different types of cells in the tissues. We also showed that the feedback on cell cycle from the growth factor is important for forming temporary ''stem cell niche'', the term that generally refers to the microenvironment where the stem cells reside and sustain their renewing functions, during the development of the tissue. Our findings are consistent with the experimental results our collaborators have observed.
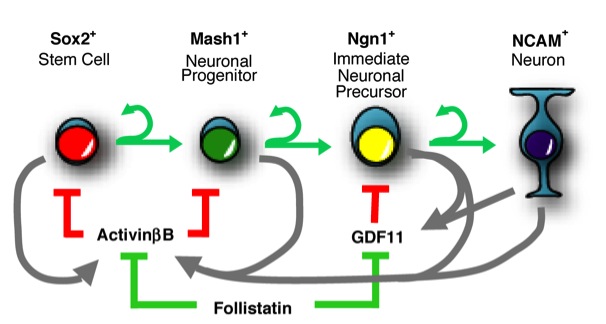
Figure: A schematic diagram of a single cell lineage and associated regulatory molecules. Cells proliferate, differentiate to the next lineage stage or undergo death. (from ref.2)
Cell polarization is essential for a variety of cellular processes and function. The small GTPase Cdc42 plays a central role in polarity establishment from yeast to humans. The budding yeast provides a unique model system to study the development of cell polarity owing to its simplicity and experimental feasibility. During vegetative growth, yeast cells choose a specific bud site depending on their cell type, and selection of a bud site determines the axis of polarized cell growth. Haploid a and a cells bud in the axial pattern, in which both mother and daughter cells select new bud sites adjacent to their immediately preceding division sites. In contrast, a/a cells (such as normal diploids) bud in the bipolar pattern, in which daughter cells usually bud at the distal pole (pole distal to the birth scar), and mother cells can choose a new bud site near the proximal pole (birth pole) or the distal pole. These different budding patterns occur in response to cell-type-specific markers. The Rsr1 GTPase module, which is composed of Rsr1 (also known as Bud1), its GTPase activating protein Bud2 and its GDP-GTP exchange factor (GEF) Bud5, links the cell-type-specific information to the downstream polarity establishment machinery including Cdc42. In response to temporal and spatial signals, Cdc42 becomes polarized at a predetermined cortical site to drive bud growth.
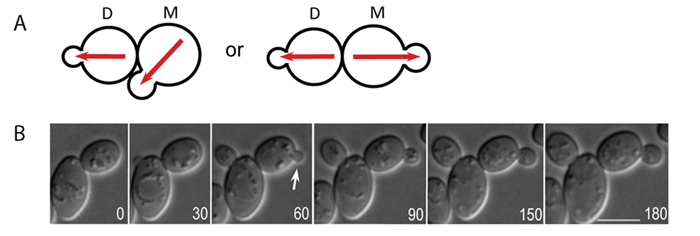
Figure: Time-lapse microscopy of Cdc42-GTP polarization in wild-type a/a diploids. A. A schematics diagram of the bipolar budding pattern. M and D stand for mother and daughter cells, respectively. Red arrows depict the axis of cell polarity. B. Time-lapse DIC image of diploid cell of wild type (YEF473). Arrows indicate budding events from daughter cells. Size bars, 5 µm. (from ref.2)
The Cdc42 GTPase plays a key role in cell polarization in budding yeast. Although previous studies in budding yeast suggested positive feedback loops whereby Cdc42 becomes polarized, these mechanisms do not include spatial cues, neglecting the normal patterns of budding. In this project, we present a two-equation reaction-diffusion model of cell polarization with a general function form of positive feedback. In the first part, we perform linear stability analysis, in particular Turing stability analysis to the model to derive conditions of parameters for which cell polarity emerges without any spatial cue. Also, we combine live cell imaging and mathematical modeling to understand how diploid daughter cells establish polarity preferentially at the pole distal to the previous division site despite the presence of two landmark cues at distal pole and proximal pole. We report that both spatial landmarks and GTP hydrolysis of Cdc42 by Rga1 controls the robust Cdc42-GTP polarization in diploid daughter cells.
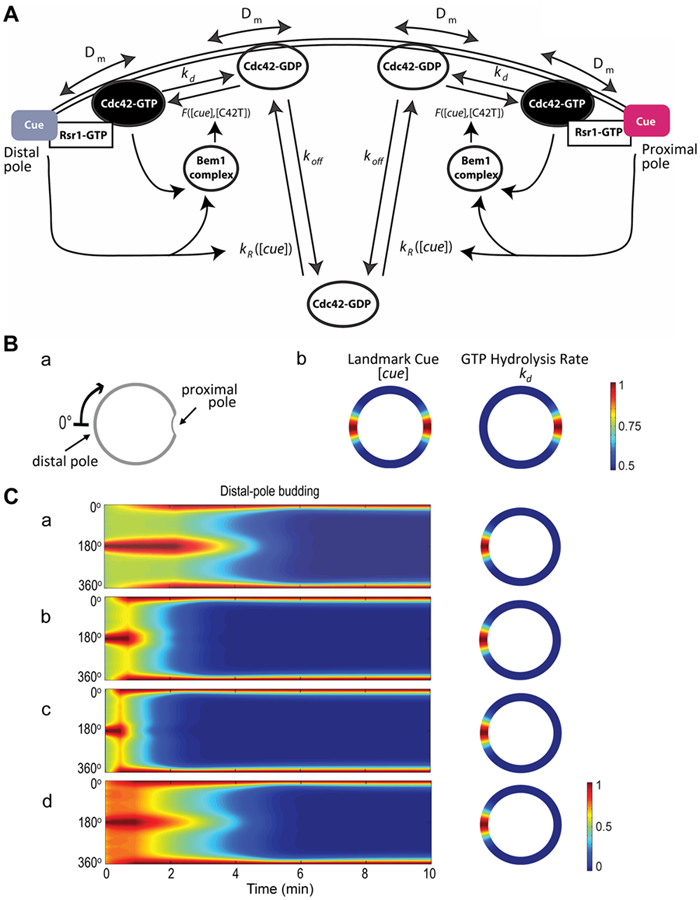
Figure: Mathematical modeling of Cdc42 polarization. A. A schematic diagram of the reaction-diffusion model with the following parameters: Dm (the diffusion rate coefficient of Cdc42-GDP and Cdc42-GTP on the plasma membrane), kd (the inactivation rate coefficient of Cdc42 from the GTP- to the GDP-bound states), and koff (the rate at which the membrane-bound Cdc42-GDP is extracted into the cytoplasm). The function F([cue], [C42T]) represents the Bem1-mediated activation rate; and the function kR([cue]) is the landmark-signal-dependent recruitment rate of Cdc42 from the cytoplasm to the membrane. Ba. Coordinate of the periphery of an a/a daughter cell. The cell periphery is parameterized by the radial angle (0°–360°) in a clockwise direction starting from the distal pole. Bb. Spatial distributions of the landmark cues and the GTP hydrolysis rate of Cdc42. Both quantities are normalized by their maximal values for better visualization, with the color map scale shown on the right. C. Spatiotemporal dynamics of Cdc42-GTP in a diploid daughter cell. The cell periphery is presented as a 1D vertical axis, with the proximal pole in the middle (180°) and distal pole at the top/bottom (0°/360°). The horizontal axis represents the time window from 0 to 10 min. The localization of Cdc42-GTP at steady state is shown on the 2D cell periphery (right). The level of Cdc42-GTP is normalized by its maximal value at steady state, and the color map is displayed. (from ref.2)
Gut mucosal homeostasis depends on complex interactions among the microbiota, the intestinal epithelium, and the gut associated immune system. A breakdown in some of these interactions may precipitate inflammation. Inflammatory bowel diseases, Crohn's disease, and ulcerative colitis are chronic inflammatory disorders of the gastrointestinal tract. Patients with ulcerative colitis or Crohn's disease are at risk of developing colorectal cancer as a result of chronic inflammation of their colon.
In this project, we constructed a model of colon cancer associated with inflammatory bowel diseases. Our model involves APC and TP53 genes, under chronic inflammation, as well as NF-?B, ß-catenin, MUC1 and MUC2. Unlike normal colon mucosa, the inflammed colon mucosa undergoes genetic mutations, affecting, in particular, tumor suppressors TP53 and APC gene. Their model demonstrated that increased level of cells with TP53 mutations results in abnormal growth and proliferation of the epithelium; further increase in the epithelium proliferation results from additional APC mutations. Since inflammatory bowel disease (IBD) is one of the critical factors in colon cancer, we developed a model of IBD. The model demonstrates how abnormal levels of T cells observed in IBD can arise from abnormal regulation of Th1 and Th2 cells by Treg cells.
The patterning of many developing tissues is organized by morphogens. Genetic or environmental perturbations in synthesis, binding, and other processes usually introduce randomness and reduce accuracy in morphogen-mediated patterning. While it has been found that the robustness of morphogen signals to perturbation of morphogen synthesis could be achieved through few particular mechanisms in certain parts of the patterning regions, how such mechanisms may affect other performance objectives of morphogen patterning, such as robustness to perturbation of receptors for the corresponding morphogen, remains a major unanswered question.
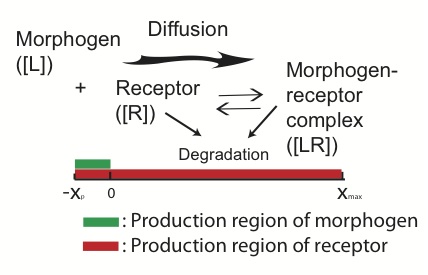
Figure: Morphogen-mediated patterning. The morphogens are produced in a local production region and diffuse toward the patterning region. The receptors are produced in the entire region and bind with the diffusive morphogens to form a morphogen-receptor gradient.
In our study, we investigate robustness of patterning to receptor synthesis in conjunction with robustness to morphogen synthesis and capability of morphogen gradient in establishing precise threshold positions, which may be measured by width of a transition zone around borders of patterns. Our analytical estimates show several tradeoffs and constraints in achieving these three performance objectives simultaneously. The explicit dependence of length scales of the patterning on the three performance objectives reveals several potential mechanisms to mediate such tradeoffs and constraints. One mechanism is through downregulation on receptor synthesis in the morphogen synthesis region and the other involves presence of non-signaling receptor molecules, both seen in the Drosophila wing disc patterning. Direct computational exploration demonstrates that these two mechanisms can improve robustness and accuracy of morphogen-mediated patterning through their ability to relax the constraints imposed by the three objective performances.
An inhomogeneous steady state pattern of nonlinear reaction–diffusion equations with no-flux boundary conditions is usually computed by solving the corresponding time-dependent reaction–diffusion equations using temporal schemes. Nonlinear solvers (e.g., Newton’s method) take less CPU time in direct computation for the steady state; however, their convergence is sensitive to the initial guess, often leading to divergence or convergence to spatially homogeneous solution. Systematically numerical exploration of spatial patterns of reaction–diffusion equations under different parameter regimes requires that the numerical method be efficient and robust to initial condition or initial guess, with better likelihood of convergence to an inhomogeneous pattern.
Our new approach that combines the advantages of temporal schemes in robustness and Newton’s method in fast convergence in solving steady states of reaction–diffusion equations is proposed. In particular, an adaptive implicit Euler with inexact solver (AIIE) method is found to be much more efficient than temporal schemes and more robust in convergence than typical nonlinear solvers (e.g., Newton’s method) in finding the inhomogeneous pattern. Application of this new approach to two reaction–diffusion equations in one, two, and three spatial dimensions, along with direct comparisons to several other existing methods, demonstrates that AIIE is a more desirable method for searching inhomogeneous spatial patterns of reaction–diffusion equations in a large parameter space.

Figure: Three-dimensional pattern computed by AIIE for Gray-Scott model. (from ref.1)